- Mateo's Rabbit Burrow
- Posts
- Is cancer a navigational problem or a destruction problem?
Is cancer a navigational problem or a destruction problem?
Versión en español
"Is cancer too much life?"
I asked myself out loud, like trying to remember an answer mid-exam.
What if cancer cells are just too good at surviving? They divide. They hide from the immune system. In tough conditions, they adapt. They switch energy sources. They find a way to stay alive when nothing else would. They are, in a brutal sense, extraordinarily good at evolving.
Few people know how much thought I have given to this problem. The more I sat with it, the more I saw that the real issue was not the unsolved answer. It was the question.
I lean on big questions. The ones too big for small talk. So I followed the thread. If cancer is too much life, what does that mean? Can we bring a cell back to its healthy state?
That question took me somewhere I did not expect.
How do you reset a system under the laws of physics?
In physics, you return a system to its lowest energy state required to stay stable. One way to do this is called annealing. You heat a disordered material. Then you cool it slowly. The material finds its natural resting state. No chaos. No force after energy injection. Just the right conditions to return.
This is one of several physical methods scientists have thought about applying to cells (see more). I keep coming back to it because of what it implies. You don't fight the disorder. You give the system what it needs to redirect.
A question followed immediately. If you pushed a cancer cell out of its current state, would it go back to a healthy state? Or does it land somewhere worse?
My honest answer: it depends on the shape of the landscape of choices around. Biology has a model for this: the Waddington landscape. Think of a ball rolling down a hill into a valley. In a healthy body, cells settle in the right valleys. In cancer, something reshapes the hill. New valleys form. The cell rolls into the wrong one and stays. The cell didn't fail. It followed the given path to the wrong place.
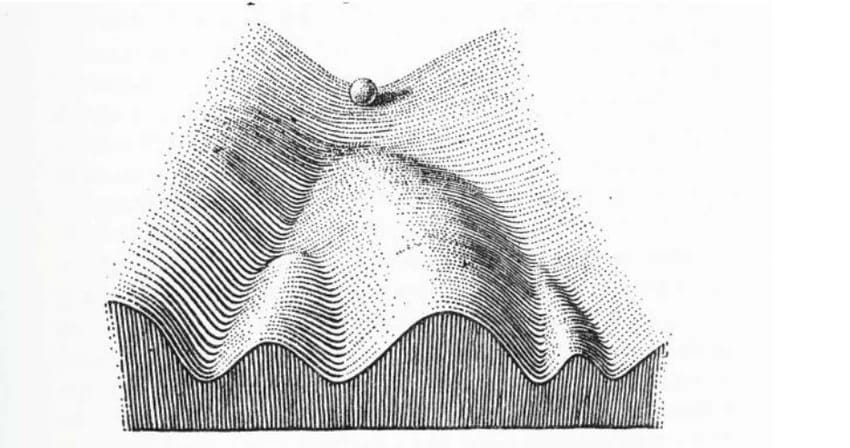
FIGURE 1: Waddington‘s landscape model. (From Waddington in 1957, 29)
Now the cell annealing question becomes: can you give the cell enough energy to climb back out? And is the right valley still reachable?
And once again, timing becomes the main variable.
Is cancer a navigational problem or a destruction problem? Is the cell lost, or too far gone to come back?
What if the problem is not damage that must be cut, poisoned, or burned? What if it is a wrong direction?
The proof already exists. Differentiation therapy. Scientists asked a simple question. What if instead of killing leukemia cells, you told them to grow up? ATRA did exactly that. In acute promyelocytic leukemia, cure rates with older anthracycline regimens were about 40%, and the introduction of ATRA (later combined with arsenic trioxide) increased cure rates to roughly 70–90% with modern combined therapy. The cell wasn't destroyed directly. It was redirected to mature and disappear. CRISPR therapies extend the same navigational logic at the genome level: change the instructions that drive disease, and you change the trajectory. Not just the body count. That is the navigational frame in action.
The alternative is what medicine defaults to when the landscape has already closed. Find it late. Kill it. Cut, poison, burn. After the cell has settled in the wrong place, the energy required to return it becomes too much. It answers a different version of the cancer problem entirely.
T cell therapy shows how the same tool works on both sides, depending on when you arrive. Early stage: checkpoint inhibitors such as PD-1 and PD-L1 don't add firepower. They remove a wrong signal. The cancer told the immune system, "Nothing to see here." The therapy corrects the lie. That is navigational. Late stage: CAR-T turbocharges T cell killing. Seek and destroy. That is demolition. Same tool. Completely different theory. The difference is not the weapon. It is when you show up.
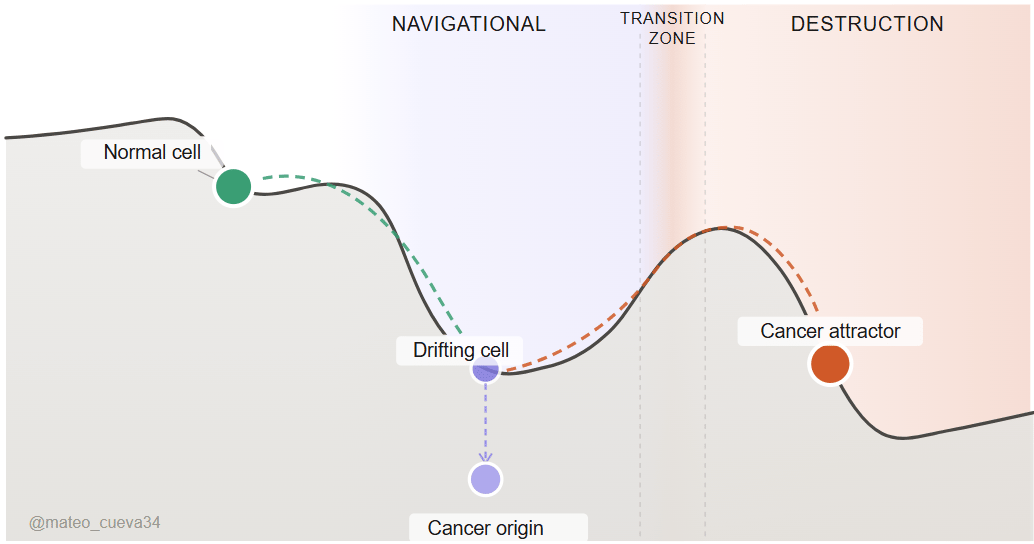

Can we find the transition from a navigational problem to a destruction problem?
The navigational stage is where the cell is still drifting, still in motion, still reachable. The destruction stage is where the landscape has closed, the cell has settled, and redirecting it requires far too much energy. The navigational stage is not fixed in time. Some cancers drift slowly over years. Others cross the line in months. The window is real, but it doesn't wait.
Finding the point of no return directly has been the nightmare and goal of early cancer detection for a long time. Liquid biopsy. Methylation panels. Circulating tumor DNA. All brilliant attempts to locate the exact moment the landscape closes. Partial results at best when separated. Not because the idea is wrong, but because the biological signal at that moment is buried in noise. The body is doing a thousand things at once.
And here is what makes it even harder. The transition point is not a single line. It is a phase transition, like water turning to steam. There is a moment of latent heat where the cell still looks navigational. But it is already energetically committed to becoming a destruction problem. The signal exists. But it does not announce itself. When approached head-on, it keeps disappearing into the data.
So why not?
Invert.
We have a better idea of what a cell in the full destruction stage looks like. Locked metabolic programs. Surface identity changes. Immune evasion fully operational. We know what a healthy cell looks like. The navigational stage is what remains when you subtract both. Not found by searching. Found by elimination.
Two filters. Map destruction signals. Map healthy signs. The intersection narrows the noise to the window that matters. What is neither healthy nor fully malignant is the target. Invert again. Do not ask what signals appear at the cancer origin. Ask what signals disappear from healthy tissue first. Methylation drift, surface protein expression changes, and immune surveillance failures. These are not the arrival of cancer. They are the departure from health.
Charlie Munger said it plainly. Invert, always invert. "All I want to know is where I'm going to die, so I'll never go there."
The navigational stage isn't where cancer announces itself. It is what remains after we have removed every other explanation.
The hardest state to observe directly is often best found indirectly.
One unsolved problem with no edges is noise.
Two problems with edges are a map.
Take care)
Mateo
Sources and papers page
I’m taking this framework from theory into a functioning workflow.
Project Calchas - intro & update 1:
Pancreatic cancer has an over 75% late-stage diagnosis. The navigational window exists. The signals exist. The noise is the problem. Current detection approaches search for the transition point directly. It keeps disappearing into the data.
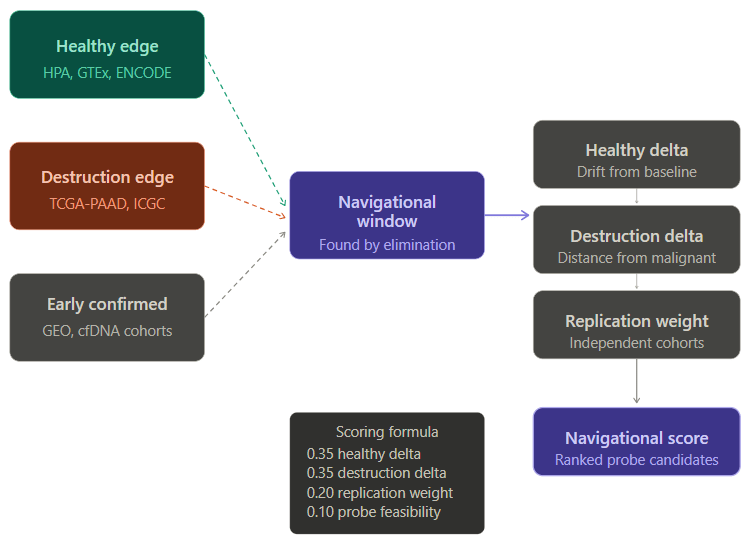
Based on the proposed framework, map edges. Subtract. Find what remains.
Left wall: healthy pancreatic tissue signals. HPA, GTEx, ENCODE.
Right wall: confirmed late-stage PDAC signals. TCGA-PAAD, ICGC.
Subtract both. Score what survives by drift from health, distance from malignancy, and replication across independent cohorts and clinical studies.
Output: ranked probe candidates; the signals most worth building around.
Next steps: MD, kinetic model, virtual cohort analysis of best candidates.
First step in a longer hypothesis. The right signals inform a dual probe molecule, self-correcting readings that amplify each other, designed to cut through what MCED panels alone cannot resolve in high-risk populations. Not a replacement for broad screening. A precision booster for the people most likely to be in the navigational window already.
The end goal: pancreatic cancer caught before the window closes, starting with the populations where catching it early matters most.
More project updates soon.